- Overview
- Course outline
- Entry
- Career path
- How to apply
Course Overview
This part-time Level 7 Bachelor of Science in Process Digitalisation is a 60 ECTS degree programme that provides a unique learner experience where data analytics meets automation, in a blended learning format. It will provide industry-ready graduates, equipped to meet current and future demands, essential to maintain modern manufacturing and smart manufacturing operations in Ireland.
The fastest-growing roles across many industries are driven by technology, digitalisation and sustainability. This increased adoption of new and frontier technologies drives the need for advanced skill sets, including specialist roles such as Business / Data Intelligence Analysts, Automation / Sustainability Specialists and Operational Excellence Professionals.
As well as completing modules in the future of manufacturing and supply chain, management of data and the concepts of validation and concepts of quality assurance, learners will understand the growing trends and compliance relating to sustainability. Additional modules will support the learner further in employment by completing modules in operational excellence, industrial automation and process control along with data analytics and data-driven project management.
Graduates will possess the skills and competencies necessary to implement digitalised processes, an area where a critical skills shortage is emerging nationally. The curriculum is designed to produce graduates capable of digitalising a process to drive improvements in their business/manufacturing or manufacturing operations while applying their knowledge of the ethics and principles of sustainability to their work.
Developed and taught by experienced academics and industry lecturers, this course is delivered through blended learning, 2 evenings per week online and up to 2 Saturdays per month in person.
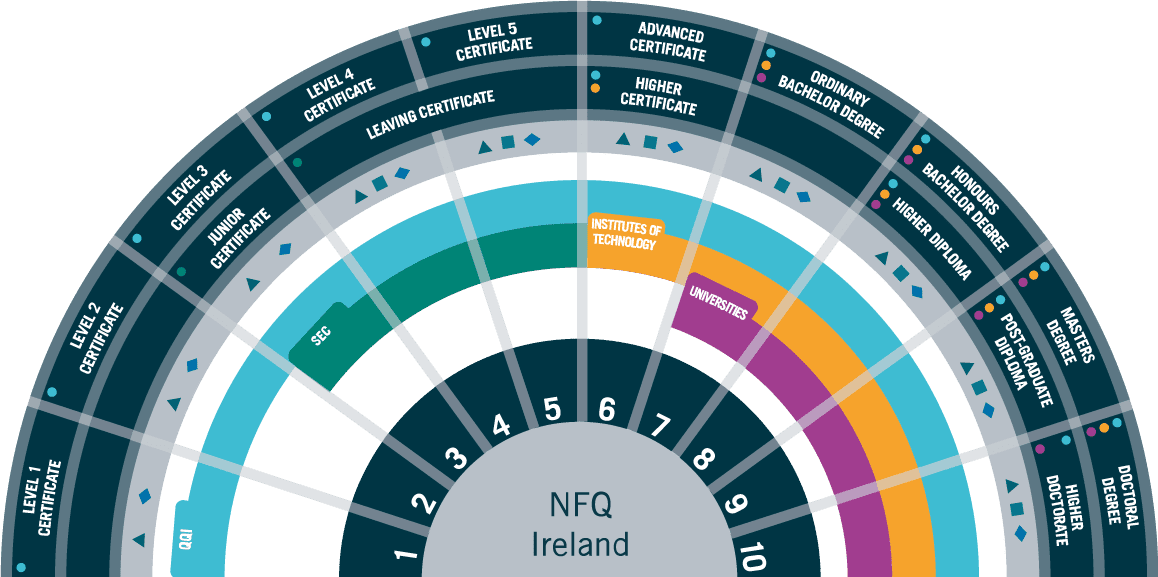
Accreditation: QQI Level 7 Bachelor of Science in Process Digitalisation

Course Outline
This part-time programme runs over 3 semesters. Lectures are delivered online 2 evenings per week plus 1-2 Saturdays per month in a face to face classroom.
This course will cover the following modules.
Module 1: The Future of Manufacturing and Supply Chain
Explore sustainable practices, evaluate supply chain impacts, and apply climate intelligence for risk monitoring.
Module 2: Progressing towards a sustainable industry
Discover sustainability concepts, including circular economy, supply chains, and digitalization, for practical application in professional careers.
Module 3: Operational Excellence in a Digital Environment
Explore traditional and updated operational excellence models in the context of digital transformation.
Module 4: Validation and the Concepts of Quality Assurance
Understand validation processes, quality management, regulatory requirements, and the role of digitalization in improving industry standards.
Module 5: The Management of Data
Learn about database management systems, relational and NoSQL data models, data manipulation, distribution, ethics, and EU legislation.
Module 6:Data-Driven Project Management
Learn data-driven project management, agile methodologies, and data visualization for effective communication and project planning.
Module 7: Industrial automation and process control
Gain skills in automation, digitalization, and process control, covering architecture, measurement, analysis, and control methods.
Module 8: The Analysis of Data
Introduction to data analysis, tools, and statistical techniques for effective decision-making in the era of big data.

Entry Requirements
Applicants are required to hold a minimum of a QQI Level 6 Certificate (120 credits) in Science, Engineering, Quality or related discipline.
Career Options
Over 70% of graduates have successfully advanced their careers post completion of our courses. Potential career opportunities following the successful completion of this course are highlighted below:
- Data Analyst
- Business Analyst
- Associate Automation Engineer
- Digital Construction Lead
- Associate Project Lead
- Junior Validation Specialist
- Automation Technician
- Process Improvement Specialist
An essential part of the course will be the career progression module where significant support will be given to participants on career development through structured one-on-one CV assistance, interviewing support and developing career networks. regular guest speakers and lecturers from the industry will assist graduates in making links with the industry.
Apply Now
Our admissions team are on hand to assist you with your application and answer any questions you may have on the course.
Students who wish to apply to this course should follow the following application process.
Step 1: Enquire through the form at the top right of this page
Step 2: A member of our team will be in contact through phone or email
Step 3: If you are deemed to be eligible for the course, you will be sent an application form by email. You MUST fill in this application form and attach all necessary *documents (CV, ID, Transcripts, etc.)
Step 4: On submission of the completed application form and all documentation, your application will be sent to the associated department for final approval. You will receive an e-mail confirming your place on the programme. (Note: This process can take up to one week particularly during busy admission periods.)
*We accept scanned documents in a pdf format. Pictures of documents are not accepted.
**For those who are applying for Springboard funding, you will be directed to fill out an additional application form on the Springboard website, this is to confirm your eligibility to receive funding
Please note at any time if you have any questions please do not hesitate to contact us by email on admissions@innopharmalabs.com or call us (01) 485 334
Why up-skill for the STEM process manufacturing sectors in Ireland?
- Over 130,000 employed within these industries
- Over 40,000 new jobs predicted
- Over €62 billion in exports
- 9 of top 10 world’s biopharma companies in Ireland
- 8 of the top 10 worlds medtech companies in Ireland
- Over 8,000 new jobs predicted

Testimonials
The course had online lectures so anytime I couldn’t go to college I could join the class online. I never missed a class.
BA in Pharmaceutical Business Operations Graduate, Joanna Galon, Senior Associate Regulatory Affairs at Amgen
Our Blog
Blog
February 9, 2026
The Open Research Revolution: Digital Transformation and the Future of Evidence-Based Learning
Author: Colm O’Connor, College Librarian & Research Specialist, Innopharma Education In today’s rapidly evolving...
Blog
February 3, 2026
Digital Transformation: Cultivate the Talent Rather Than Buy the Platform
Author Finbarr Sheehy Digital transformation is often sold as a technology story: choose the right platform, implement it well and...
Blog
December 12, 2025
The Life Sciences Workforce Is Changing — Are You Ready?
AI is no longer a futuristic idea hovering on the horizon — it’s already woven into the daily reality of pharma, biopharma, an...






















