- Overview
- Course outline
- Entry
- Career path
- How to apply
Course Overview
This internationally recognised online Masters in Pharmaceutical Business & Technology is a unique blend of technology and business skills.
This programme consists of modules about the processes of pharmaceuticals and their production, as well as infusing your knowledge of the business and technical side of the sector. This will help give you an extra edge in your qualification. In addition, you’ll be awarded insights into the newly emerging trends of the industry, along with classes in leadership development and strengthening your professional skills.
With extensive input and participation from the pharmaceutical and clinical trials sectors, this course is ideal for those involved in manufacturing, compliance, business improvement, quality assurance, validation, data analytics, clinical trial coordination and engineering.
Domestic & EU applicants – With its unique online part-time delivery, this course is flexibly designed to suit the needs of those in employment. Classes are broadcast online, offering the learners the opportunity to join from home. Students are required to attend on-campus 2 Saturdays per month.
International applicants – Full-time study is available for international students. The full-time course duration is 12 months and in-class lecturers take place Monday – Friday.
Designed and delivered by highly experienced academic and industry professionals, we have developed the course content focusing on the skills companies are looking for and to advance your career.
Accreditation: QQI Master of Science (MSc) in Pharmaceutical Business & Technology.
Collaborating College: Griffith College Dublin
Next Intake: Summer 2025. Now accepting applications.


Course Outline
Online lectures take place two evenings per week with in-class lectures taking place on two Saturdays per month in Griffith College Dublin. (ECTS: 90 Credits)
A full-time option is available for International Students.
Module 1: Clinical Research Management
Drug development life cycle, clinical trials project management, contract research organisations (CRO), competitive marketing in the clinical trials sector, and managing commercial relationships.
Module 2: Processes, Production and Pharmaceutical Quality Systems
Pharmaceutical & Biopharmaceutical manufacturing industry, pharmaceutical products, manufacturing, quality system elements, validation and change control, pharmaceutical plant design, pharmaceutical quality systems, and principles of quality by design.
Module 3: Pharmaceutical Technology Transfer
The technology transfer process, research to manufacturing, location to location, life cycle studies, raw material impacts, pilot plan studies, validation, commissioning and cleaning, project management, regulatory framework, and analytical methods.
Module 4: Regulatory Landscape of Pharmaceutical Business
Pharmaceutical legislation and regulation, core principles, statutory frameworks, environmental regulations, Irish and European Union law, planning law, waste management, control of pollution, and factory facilities.
Module 5: 21st Century Dynamics & Emerging Trends
Challenges facing the pharmaceutical industry, new product types, patent cliffs, emerging markets, innovative medicines, live licensing, modes of healthcare delivery, governmental strategies, and demands on manufacturing and distribution.
Module 6: Strategy, Leadership & the Culture of Innovation
Current challenges of the pharmaceutical industry, strategic thinking in the Biopharmaceutical industry, leading change in the biopharmaceutical industry, culture of innovation, corporate governance and business ethics.
Module 7: Operational Excellence & the Science of Innovation
Origins of operational excellence, operational excellence implementation models, lean sigma implementation, design of six sigma, science of innovation, TRIZ, key performance indicators (KPI), and quality function deployment.
Module 8: Research Methods
Topic selection, literature review, research design, methods of data collection and analysis, ethics
Module 9: Dissertation
A substantial focus of the programme will also develop the professional skills of the graduate to enhance their future career opportunities through the development of their network, presentation and communication skills

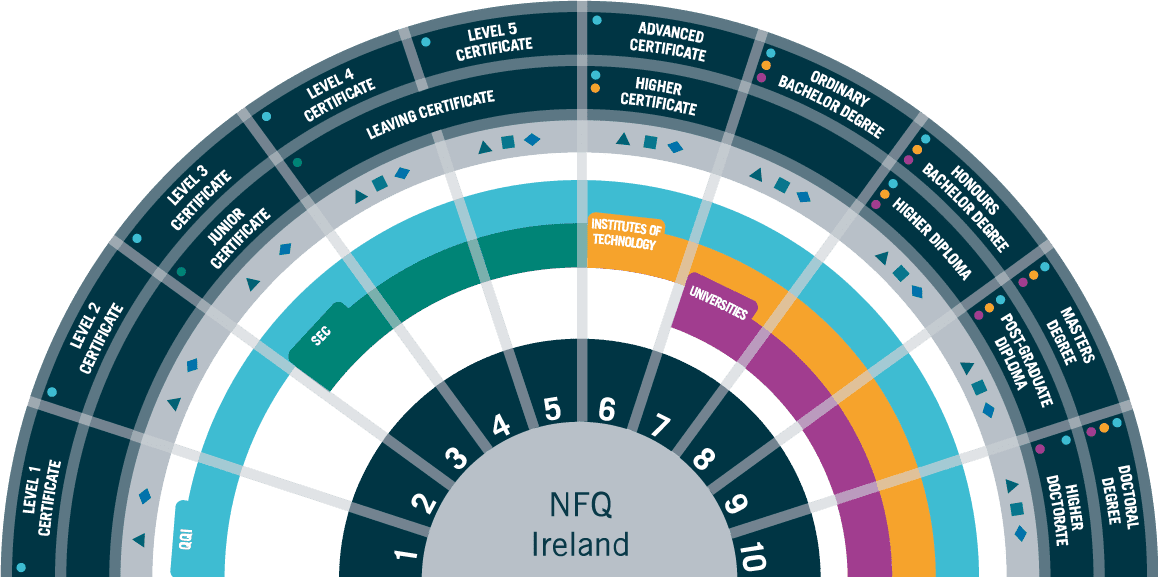
Entry Requirements
This course is suitable for those who possess qualifications at NFQ Level 8 Honours degree with a 2.2 or higher in a Science, Engineering or Quality discipline.
Career Options
This highly desired qualification will help you open the door to an exciting and successful career in the pharma or life sciences industries. This unique masters’ qualification could help kick-start your career progression as an executive, manager or director in the following specialities:
- Business Operations Supervisor/ Manager
- Quality Assurance Supervisor/Manager
- Compliance Specialist / Manager
- Business Improvement Specialist
- Clinical Trials Coordinator
- Technology Specialist/ Manager
- Quality Supervisor/ Manager
- New Business Introduction Specialist
Apply Now
Our admissions team are on hand to assist you with your application and answer any questions you may have on the course.
Students who wish to apply to this course should follow the following application process.
Step 1: Enquire through the form at the top right of this page
Step 2: A member of our team will be in contact through phone or email
Step 3: If you are deemed to be eligible for the course, you will be sent an application form by email. You MUST fill in this application form and attach all necessary *documents (CV, ID, Transcripts, etc.)
Step 4: On submission of the completed application form and all documentation, your application will be sent to the associated department for final approval. You will receive an e-mail confirming your place on the programme. (Note: This process can take up to one week particularly during busy admission periods.)
*We accept scanned documents in a pdf format. Pictures of documents are not accepted.
**For those who are applying for Springboard funding, you will be directed to fill out an additional application form on the Springboard website, this is to confirm your eligibility to receive funding
Please note at any time if you have any questions please do not hesitate to contact us by email on admissions@innopharmalabs.com or call us (01) 485 334
Why up-skill for the pharmaceutical industry in Ireland
- €3 billion in new capital investment
- €39 billion in exports
- Over 55,000 employed
- 9 of top 10 world’s biopharma companies in Ireland
- 12 of the top-selling drugs made in Ireland
- Over 8,000 new jobs predicted

Get in touch
Innopharma Education
Ravenscourt Campus, Three Rock Road,
Sandyford, Dublin 18, Ireland
Phone:
+353 1 485 3346
Have a question?
Frequent Asked Questions
The Masters is delivered over three semesters full time and five semesters part-time. Part-time learners attend classes two evenings per week and two Saturdays per month for tutorial and group work. Evening classes are delivered from Griffith College Dublin and are also broadcast live. Full-time students attend four days per week in Griffith College Dublin.
The deadline for applications is late August. The course will begin in September however lectures are recorded for those who join late. We also have a second intake in February.
A level 8 primary degree with a 2.2 or higher in Science, Quality or Engineering related discipline. Recognition of Prior Learning can also be considered to gain access.
Career opportunities include roles in operations management, technology transfer, regulatory affairs/compliance, operations excellence, clinical trials, coordination, quality assurance and business strategy. Companies in Cork, Limerick, Waterford, Tipperary, Dublin & Galway are all searching for people to fill these roles.
Our alumni are working in some of the top pharmaceutical companies in Ireland including Pfizer, Amgen, Gilead Pharmaceutical, Forest Laboratories, Allergen, Johnson & Johnson, MSD, Novartis, Sanofi, AbbVie and GlaxoSmithKline to name a few.
Yes, we accept company sponsorship.
Our Blog
Blog
April 3, 2025
AI in Biopharma: Transforming Manufacturing & Innovation
How AI, Machine Learning, and Automation are Transforming Biopharmaceutical Manufacturing Here at Innopharma, we are proud to show...
Blog
December 11, 2024
Looking for a Career in the Biopharmaceutical Industry? Everything You Need to Know
The biopharmaceutical industry is a rapidly growing sector that combines cutting-edge biotechnology with pharmaceutical science to...
Blog
October 18, 2024
What is Pharmaceutical Manufacturing & Why is it Important?
Pharmaceutical manufacturing is the name given to the production of medicinal products on an industrial scale. Pharmaceutical/Biot...






















